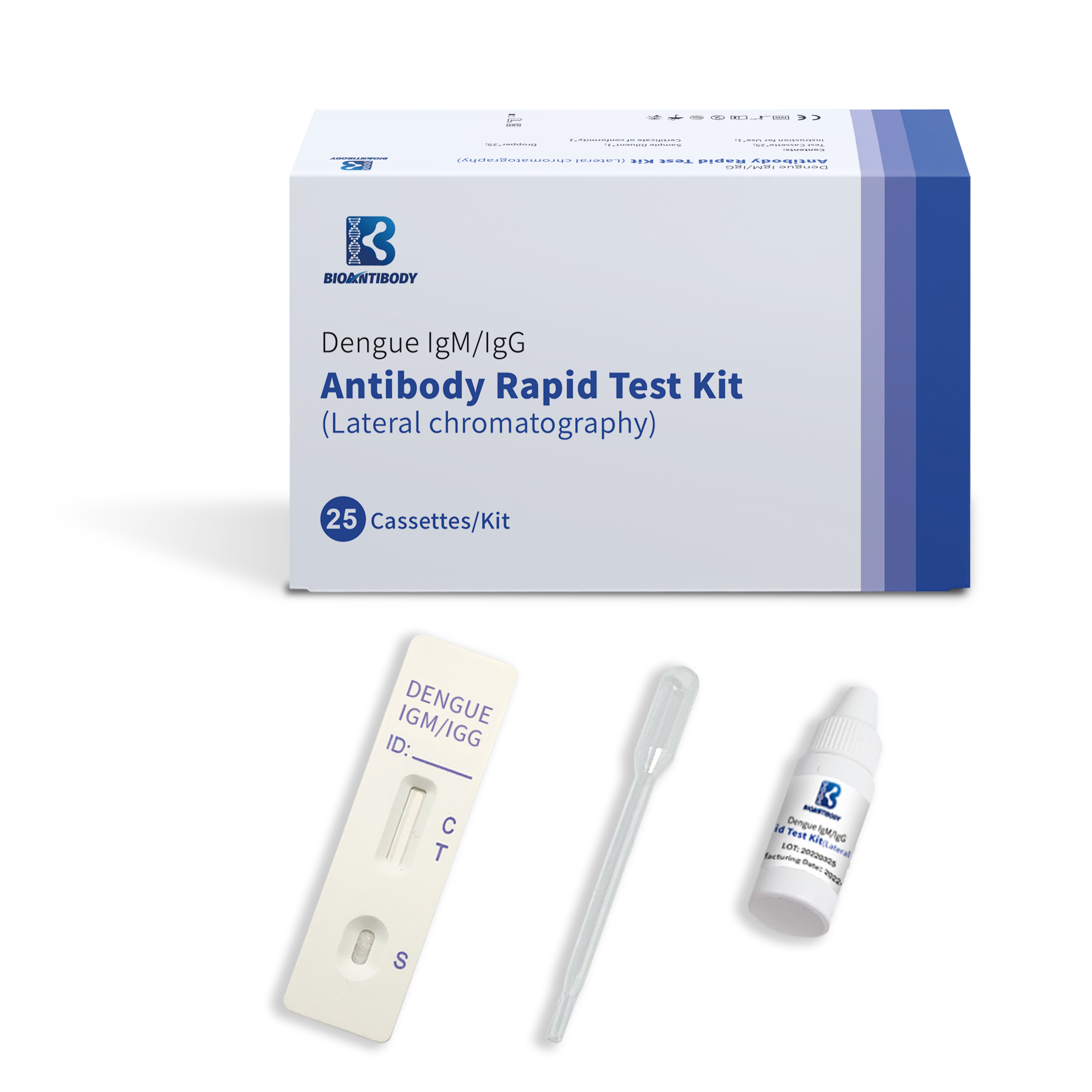
Dengue IgM/IgG Antibody Rapid test kit (Lateral chromatography)
Intended Use
Dengue IgM/IgG Antibody Rapid Test Kit (Lateral chromatography) is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG and IgM antibodies to dengue virus in human serum, plasma, whole blood or fingertip whole blood. This test provides only a preliminary test result. The test is only to be used by medical professionals.
Test Principle
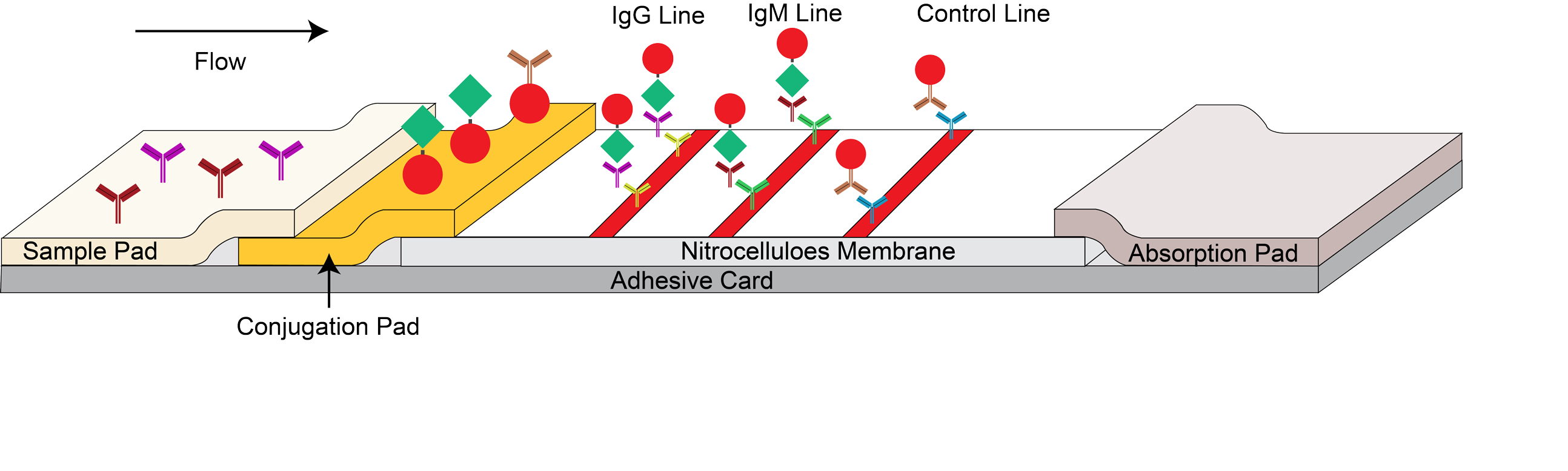
The Dengue IgM/IgG test device has 3 pre-coated lines, "G" (Dengue IgG Test Line), "M" (Dengue IgM Test Line) and "C" (Control Line) on the surface of the membrane. The "Control Line" is used for procedural control. When a specimen is added to the sample well, anti-Dengue IgGs and IgMs in the specimen will react with recombinant dengue virus envelope proteins conjugates and forms a complex of antibodies antigen. As this complex migrates along the length of the test device by capillary action, it will be captured by the relevant anti-human IgG and or anti-human IgM immobilized in two test lines across the test device and generate a colored line. Neither the test line nor control line are visible in the result window prior to applying the specimen. A
visible control line is required to indicate the result is valid.
Main Contents
Components provided are listed in the table.
| Component \ REF | B009C-01 | B009C-25 |
| Test Cassette | 1 test | 25 tests |
| Sample Diluent | 1 bottle | 25 bottles |
| Dropper | 1 piece | 25 pcs |
| Disposable lancet | 1 piece | 25 pcs |
| Instructions For Use | 1 piece | 1 piece |
| Certificate of Conformity | 1 piece | 1 piece |
Operation Flow
Step 1: Sampling
Collect human Serum/Plasma/Whole blood properly.
Step 2: Testing
1. Remove an extraction tube from the kit and a test box from the film bag by tearing the notch. Put them on the horizontal plane.
2. Open the inspection card aluminum foil bag. Remove the test card and place it horizontally on a table.
Use a disposable pipette, transfer 10μL serum/or plasma/or 20μL whole blood into the sample well on the test cassette.
Step 3: Reading
10 mins later, read the results visually. (Note: do NOT read the results after 15 mins!)
Result Interpretation
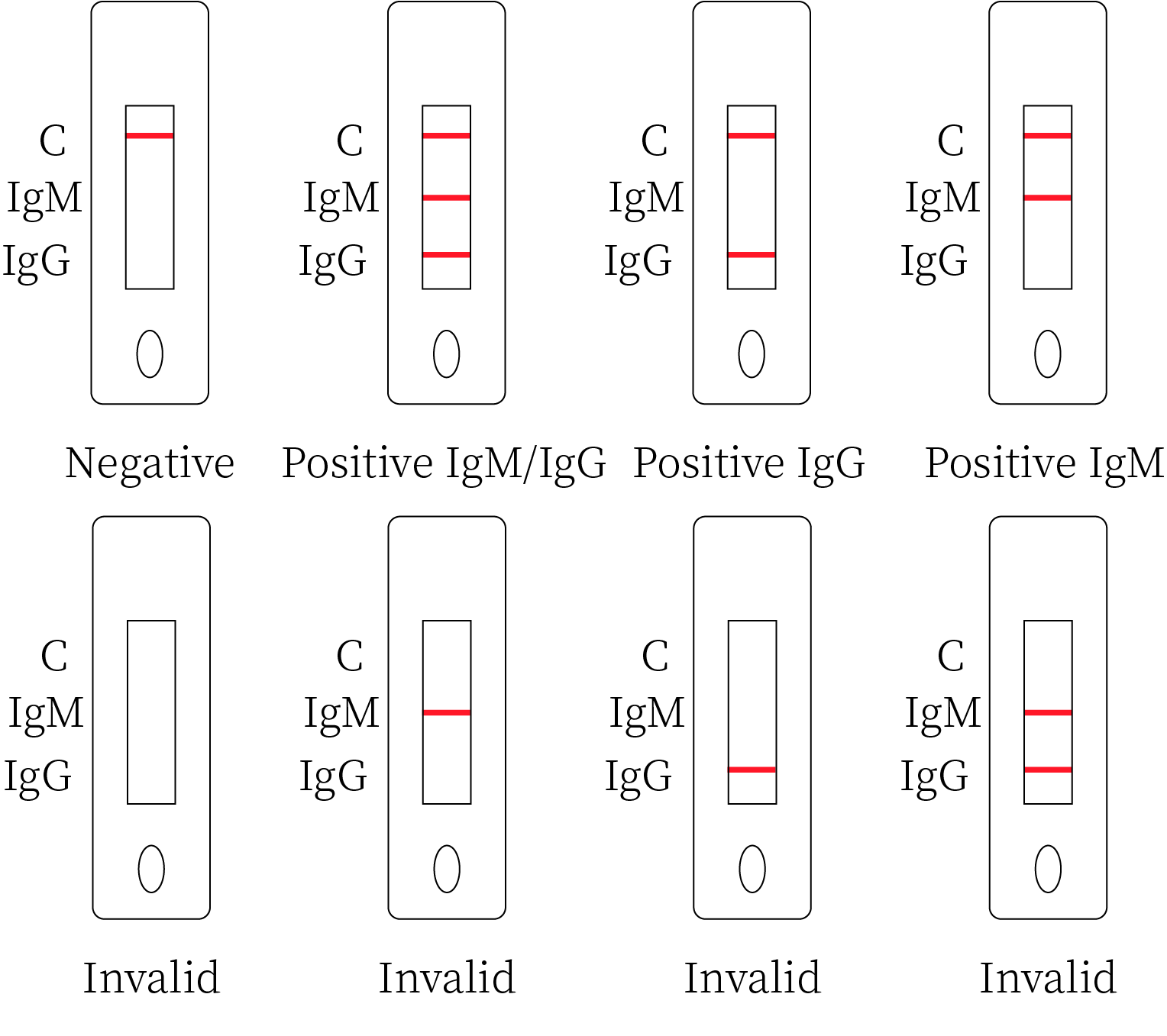
1. Positive IgM Result The control line (C) and IgM line (M) are visible on the test device. This is positive for IgM antibodies to Dengue virus. It is indicative of a primary dengue infection.
2.Positive IgG Result The control line (C) and IgG line (G) are visible on the test device. This is positive for IgG antibodies. It is indicative of secondary or previous dengue infection.
3. Positive IgM and IgG Result The control line (C), IgM (M) and IgG line (G) are visible on the test device. This is positive for both IgM and IgG antibodies. It is indicative of a late primary or early secondary dengue infection.
4.Negative Result The control line is only visible on the test device. It means that No IgG and IgM antibodies were detected.
5.Invalid Result No visible colored band appears at control line after performing the test. Insufficient sample volume or incorrect procedural techniques are the most likely reasons for control line failure. Review the test procedure and repeat the test using a new test device.
Order Information
| Product Name | Cat. No | Size | Specimen | Shelf Life | Trans. & Sto. Temp. |
| Dengue IgM/IgG Antibody Rapid test kit (Lateral chromatography) | B009C-01 | 1test/kit | Serum/Plasma/Whole Blood | 18 Months | 2-30℃ / 36-86℉ |
| B009C-25 | 25 tests/kit |